To compare the columns used, numerous chromatographic tests were evaluated. The establishment of similarities or dissimilarities between particular stationary phases as well as the description of the retention mechanism induce the validity of such research for both groups. The practitioners and theoreticians of HPLC are interested in the chromatographic evaluation of new and commercial chromatographic packings. have also demonstrated differences between linear and cyclic tetrapeptide-bonded stationary phases. developed hydrophilic tripeptide-based organosilica hybrid stationary phases dedicated for analysis of bioactive polar compounds under HILIC and RP conditions. applied in off-line two-dimensional liquid chromatography (2D-RP/RP LC). These materials were evaluated anew by Xue et al. di-, tri-, and tetrapeptides) were also investigated with regard to chemically bonded stationary phases for LC.

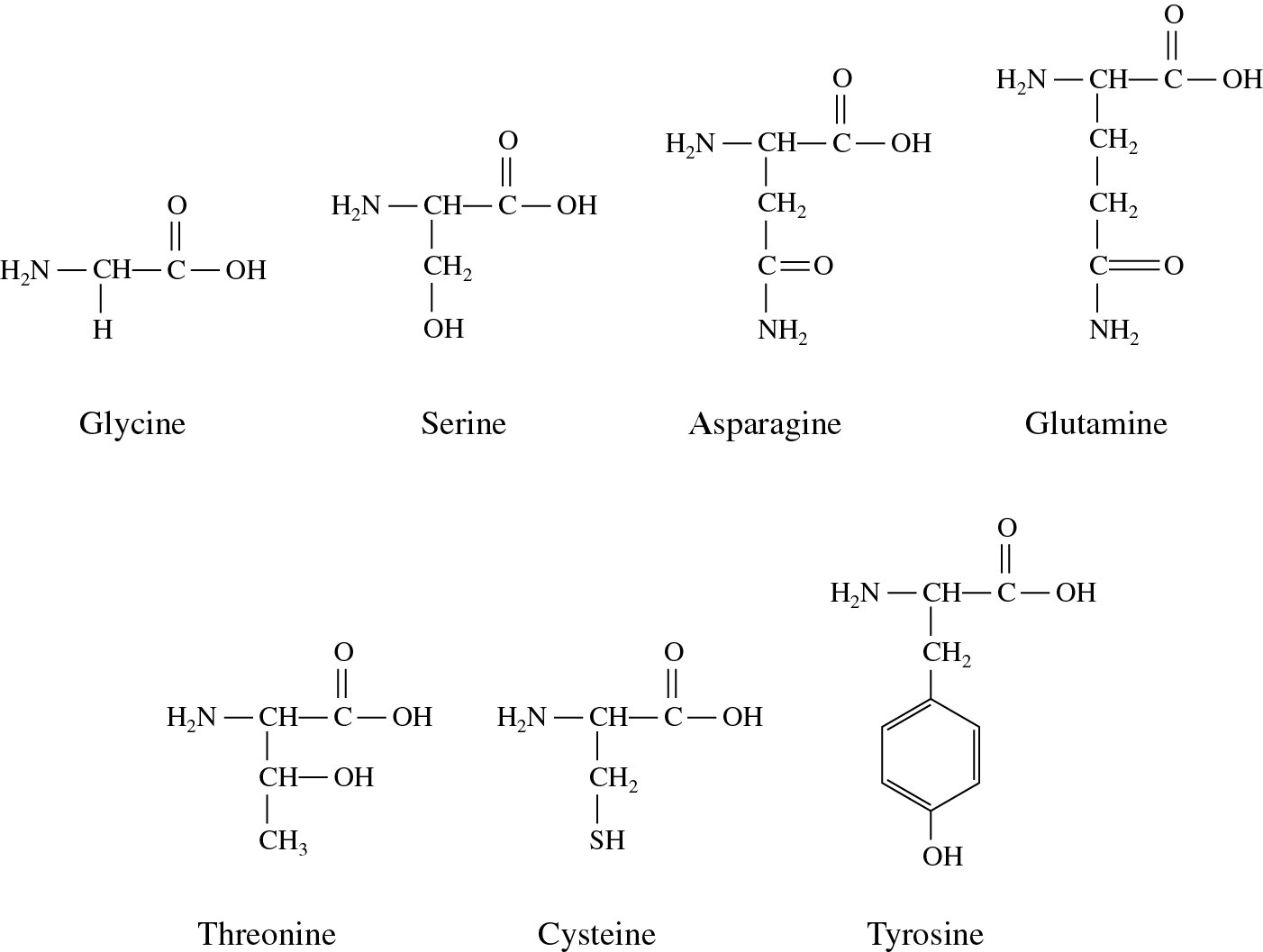
On the other hand, polypeptides as chemically bonded ligands were evaluated in terms of enantioselectivity. Amino acids are natural zwitterionic compounds and their immobilization on the silica surface enables the preparation of ion-exchange stationary phases for both HILIC and ion chromatography (IC) systems. In the last decade, there have been several investigations of amino acids, peptides, and polypeptides grafted onto silica as stationary phases for liquid chromatography (LC). Peptide-bonded ISRP packings were specifically designed for separation of drugs in blood serum or plasma by direct injection. Peptide-bonded phases were also an essential component of the internal surface reversed-phase (ISRP) columns introduced by Hagestam and Pinkerton. Grushka and Scott the originators of this type of materials, applied bonded optically active tripeptide stationary phases for the separation of d, l-amino acids, isomeric dipeptides, and phenyltiohydantoin (PTH)-amino acids derivatives. Depending on the immobilized sequence of amino acids, these materials exhibit diversified application targets. Recently according to impressive progress in hydrophilic interaction chromatography (HILIC), evaluation of stationary phases with, inter alia, chemically bonded amino acids and peptides could be observed. Linear peptides constitute convenient organic components in separation materials due to a wide range of side chain chemistries (linear, branched, or aromatic alkyl groups, thiols, amines, carboxylic acids, etc.). The performed research assisted in the evaluation of the relation between the chemical properties of bonded amino acid sequence and their role in the retention mechanism. The factor analysis (FA) based on the principal component analysis (PCA) was also used in the stationary phase comparison. The results of the selectivity for partial structural differences were presented as radar-shaped plots and in tabular format. Under RP HPLC conditions, following features were elucidated: hydrophobicity, hydrophobic selectivity, shape selectivity, hydrogen bonding, electrostatic interactions at pH values 2.7 and 7.6. In the case of HILIC mode, the characterization was performed in terms of the degree of hydrophilicity, selectivity for hydrophilic–hydrophobic compounds, selectivity for the regio and configurational substituents, anion and cation exchange properties, and acidic or basic nature of the stationary phase surface. Desired interactions were derived from the retention variation between pairs of similar solutes differing with particular structure properties. The investigated stationary phases were modified by the following amino acids: phenylalanine, leucine, alanine, glycine, and aspartic acid. The comparison between 9 home-made amino acid- and peptide-silica stationary phases in hydrophilic interaction liquid chromatography (HILIC) and reversed-phase liquid chromatography (RP HPLC) systems has been presented. Amino acids and peptides (di- and tripeptides) as chemically bonded ligands in liquid chromatography were investigated.
